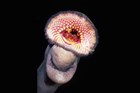
Pouch Lamprey, Geotria australis Gray 1851

A Pouch Lamprey, Geotria australis. Source: Rudie H. Kuiter / Aquatic Photographics. License: All rights reserved
Primitive eel-like fishes with two dorsal fins near the tail, 7 pairs of pore-like gill openings, a mouth in the shape of an oral disc fringed with skin flaps and armed with many small, horny teeth, including spatulate lateral teeth, and eyes positioned on the side of the head.
Pouch Lampreys have a cartilaginous skeleton, primitive eyes, a single median nostril, and lack true jaws, paired fins and scales. Breeding males have a large baggy pouch on the underside of the head.
These jawless fishes are anadromous - adults spawn in the headwaters of freshwater rivers and streams, and when the larvae or ammocoetes hatch, they drift downstream and burrow into soft muddy sediments. They spend the next few years filter-feeding on micro-organisms from the water above.
After metamorphosis, young adults migrate downstream to estuaries and coastal waters, where they feed parasitically by rasping flesh from other fishes with their toothy tongues. They eventually cease feeding and migrate back to freshwater to breed.
Video of Pouched Lamprey in south Western Australia
Pouch Lamprey, Geotria australis Gray 1851
More Info
|
Distribution |
Marine, estuarine and coastal rivers and streams on the south coast, from the Gippsland Lakes, Victoria, to the Moore River north of Perth, Western Australia, including coastal rivers and streams in Tasmania. Elsewhere the species is widespread in cold temperate waters of the Southern Hemisphere: southern Australia, New Zealand, South America, Falkland Islands, and South Georgia. Pouched Lampreys are anadromous, with adults spawning in freshwater. The worm-like ammocoete larvae live in muddy burrows in the upper reaches of coastal streams for about 4 years, before migrating downstream as young adults to spend about two years in the Southern Ocean, before returning to freshwaters to breed. |
|
Features |
Body eel-like with 7 pore-like gill openings, a single median nostril on top of the head, and eyes positioned laterally on side of head. The mouth lacks jaws, and is modified into an oral disc with many blunt, overlapping, flat, horny teeth in curved rows and several larger central teeth. The oral disc is fringed by short, fleshy papillae. Mature males have an enlarged oral disc and sometimes a baggy pouch beneath the head. Two dorsal fins are set well back near the tail, the first short-based and the second slightly longer, ending near the origin of caudal fin. Scales, pectoral, ventral and anal fins are absent. Ammocoete larvae lack eyes and have a single undivided dorsal fin. |
|
Size |
Ammocoete larvae to ~10cm; adults to 60cm TL. |
|
Colour |
Adults are dark brown to grey, and the ammocoetes are brown. Young adults at sea are silver or silvery cobalt blue, with two greenish or blue-green dorsal stripes. |
|
Feeding |
The larvae or ammocoetes begin their life as filter feeders, living mostly on detritus and microscopic algae. After metamorphosis, southern lampreys become parasitic. They use their sucker-like mouth and horny rasping teeth to attach to other fishes and tear into the flesh of their prey. Adults stop feeding when they return to freshwater rivers to spawn, surviving on stored fats and body tissues during their migration. |
|
Biology |
Newly hatched ammocoete larvae drift downstream and eventually burrowing into muddy substrates. After 4-5 years, the ammocoetes metamorphose into young adult lampreys over a period of about six months. During this time their eyes become larger and develop the visual pigments essential for life at sea. They migrate downstream, leaving behind their life as filter-feeders to become parasites on other fishes. They spend the next two years or so in the Southern Ocean before undertaking a long migration of up to 18 months back to the headwaters of freshwater streams to breed. Remarkably, the lampreys do not feed during this time. Once in freshwater, they lose their brilliant colour, becoming a drab brown. Males may also develop the large characteristic pouch on the underside of the head behind the mouth. Adults usually spawn in small, shallow, gravel-bottomed streams. The female prepares a small depression or nest in the substrate and attaches herself to a rock with her oral disc. The male attaches to the female with his oral disc and wraps around her to squeeze out her eggs before releasing his sperm. Adults die shortly after spawning. |
|
Conservation |
Lamprey populations are affected by the regulation of rivers including the construction of barriers such as dams and weirs. regulation and construction of barriers (barrages, weirs and dams).Climate change and predation by invasive species such as trout also deleteriously affect lamprey populations. |
|
Remarks |
When lampreys migrate upstream to spawn they must often negotiate obstacles such as rapids, waterfalls and even dams. Pouched lampreys are able to climb structures by gripping onto rocks and other surfaces with their sucking mouths, gradually inching their way upstream. They can even move over land to make their way upstream. |
|
Etymology |
The specific name australis (= southern) in reference to the type locality in Australia. |
|
Species Citation |
Geotria australis Gray 1851, List fish Brit. Mus. Part I: 142, Pl. 1 (fig. 3), Pl. 2. Type locality: Hobsons Bay, Victoria, or Onkaparinga, South Australia. |
|
Author |
Bray, D.J. & Gomon, M.F. 2020 |
|
Resources |
Pouch Lamprey, Geotria australis Gray 1851
References
Allen, G.R. 1982. Inland Fishes of Western Australia. Perth : Western Australian Museum 86 pp. 6 figs 20 pls.
Allen, G.R. 1989. Freshwater Fishes of Australia. Neptune, New Jersey : T.F.H. Publications 240 pp., 63 pls.
Allen, G.R., Midgley, S.H. & Allen, M. 2002. Field Guide to the Freshwater Fishes of Australia. Perth : Western Australian Museum 394 pp.
Baker, C. J., Jellyman, D. F., Reeve, K., Crow, S., Stewart, M., Buchinger, T. &Li, W. 2017. First observations of spawning nests in the pouched lamprey (Geotria australis). Canadian Journal of Fisheries and Aquatic Sciences 74: 1603-1611.
Bartels, H. & Potter, I.C. 2004. Cellular composition and ultrastructure of the gill epithelium of larval and adult lampreys: implications for osmoregulation in fresh and seawater. Journal of Experimental Biology 207: 3447–3462. doi: 10.1242/jeb.01157
Bice, C., Gorski, K., Closs, G., Franklin, P., David, B., West, D, Crow, S., Allibone, R, Ling, N. & Hitchmough, R. 2019. Geotria australis. The IUCN Red List of Threatened Species 2019: e.T197275A123377152. https://dx.doi.org/10.2305/IUCN.UK.2019-3.RLTS.T197275A123377152.en. Downloaded on 18 May 2020.
Bird, D.J. & Potter, I.C. 1983. Proximate composition at various stages of adult life in the Southern Hemisphere lamprey, Geotria australis Gray. Comp. Biochem. Physiol. 74 A(3): 623-633.
Cadwallader, P.L. & Backhouse, G.N. 1983. A Guide to the Freshwater Fish of Victoria. Melbourne : F.D. Atkinson Government Printer 249 pp. figs.
Castelnau, F.L. de 1872. Contribution to the ichthyology of Australia. 1. The Melbourne fish market. Proceedings of the Zoological and Acclimatisation Society of Victoria 1: 29-242 1 pl. (as Yarra singularis and Neomordacia howittii)
Chilcott, S.J. & Humphries, P. 1996. Freshwater fish of northeast Tasmania with notes on the dwarf galaxias. Records of the Queen Victoria Museum 103: 145-149.
Collin, H.B. & Collin, S.P. 2000. The corneal surface of aquatic vertebrates: microstructures with optical and nutritional function? Phil. Trans. R. Soc. Lond. B 355(1401): 1171-1176.
Collin, S.P., Hart, N.S., Shand, J. & Potter, I.C. 2003. Morphology and spectral absorption characteristics of retinal photoreceptors in the southern hemisphere lamprey (Geotria australis). Vis. Neurosci. 20(2): 119-130.
Collin, S.P., Potter, I.C. & Braekevelt, C. 1999. The ocular morphology of the southern hemisphere lamprey Geotria australis Gray, with special reference to the characterisation and phylogeny of photoreceptor types. Brain Behav. Evol. 54: 96-118, https://doi.org/10.1159/000006616
Cook, R.D., Potter, I.C. & Hilliard, R.W. 1990. Morphology and innervation of the buccal glands of the Southern Hemisphere lamprey, Geotria australis. Journal of Morphology 206(1): 133-145.
Davies, W.L., Collin, S.P. & Hunt, D.M. 2009. Adaptive gene loss reflects differences in the visual ecology of basal vertebrates. Molecular Biology and Evolution 26(8): 1803-1809.
Davies, W., Cowing, J., Carvalho, L., Potter, I., Trezise, A., Hunt, D. & Collin, S. 2007. Functional characterization, tuning, and regulation of visual pigment gene expression in an anadromous lamprey. FASEB J. 21: 2713–2724.
Fernholm, B. 1990. Petromyzontidae, pp. 79-80 in Gon, O. & Heemstra, P.C. (eds) Fishes of the Southern Ocean. Grahamstown : J.L.B. Smith Institute of Ichthyology 462 pp.
Fletcher, L.N., Coimbra, J.P., Rodger, J., Potter, I.C., Gill, H.S., Dunlop, S.A. & Collin, S.P. 2014. Classification of retinal ganglion cells in the southern hemisphere lamprey Geotria australis (Cyclostomata). Journal of Comparative Neurology 522(4): 750-771.
Galloway, R., Potter, I.C., Macey, D.J. & Hilliard, R.W. 1987. Oxygen consumption and responses to hypoxia of ammocoetes of the Southern Hemisphere lamprey Geotria australis. Fish Physiology and Biochemistry 4(2): 63-72.
Gill, H.S., Renaud, C.B., Chapleau, F., Mayden, R.L. & Potter I.C. 2003. Phylogeny of living parasitic lampreys (Petromyzontiformes) based on morphological data. Copeia 2003(4): 687-703.
Glover, C.J.M. 1994. Families Petromyzontidae, Myxinidae, Hexanchidae, Heterodontidae. pp. 82-90 figs 17-26 in Gomon, M.F., Glover, C.J.M. & Kuiter, R.H. (eds). The Fishes of Australia's South Coast. Adelaide : State Printer 992 pp. 810 figs.
Gomon, M.F. 2008. Families Geotriidae and Mordaciidae. pp. 28-29 in Gomon, M.F., Bray, D.J. & Kuiter, R.H. (eds). Fishes of Australia's Southern Coast. Sydney : Reed New Holland 928 pp.
Gray, J.E. 1851. List of the Specimens of Fish in the Collection of the British Museum. Chondropterygii. London : British Museum 160 pp., 2 pls.
Günther, A. 1872. Report on several collections of fishes recently obtained for the British Museum. Proceedings of the Zoological Society of London 1871(3): 652-675 pls 53-70 (as Geotria allporti)
Gustafsson, O.S.E., Collin, S.P. & Kröger, R.H.H. 2008. Early evolution of multifocal optics for well-focused colour vision in vertebrate.s. J. Exp. Biol. 211: 1559-1564.
Hammer, M.P., Adams, M. & Foster, R. 2012. Update to the catalogue of South Australian freshwater fishes (Petromyzontida & Actinopterygii). Zootaxa 3593: 59-74.
Hammer, M.P. & Walker, K.F. 2004. A catalogue of South Australian freshwater fishes, including new records, range extensions and translocations. Transactions Royal Society of South Australia 128(2): 85-97.
Hardisty, M.W. & Potter, I.C. 1971. The general biology of adult lampreys. In : Hardisty, M.W. & Potter, I.C. (eds) The Biology of Lampreys Vol. 1 (Academic Press : London)
Hardisty, M.W., Potter, I.C. & Hilliard, R.W. 1986. Gonadogenesis and sex differentiation in the Southern Hemisphere lamprey, Geotria australis Gray. J. Zool. Lond. (A) 209: 477-499.
Hilliard, R.W., Potter, I.C. & Macey, D.J. 1985. The dentition and feeding mechanism in adults of the Southern Hemisphere lamprey Geotria australis Gray. Acta Zoologica 66: 159-170.
Hubbs, C.L. & Potter, I.C. 1971. Distribution, phylogeny and taxonomy. pp. 1-65 figs 1-112 in Hardisty, M.W. & Potter, I.C. (eds). The Biology of Lampreys. London : Academic Press Vol. 1
Jellyman, D.J. & Glova, G.J. 2002. Habitat use by juvenile lampreys (Geotria australis) in a large New Zealand river. New Zealand Journal of Marine and Freshwater Research 36: 503–510.
Kelso, J.R.M. 1993. Substrate selection by Geotria australis ammocoetes in the laboratory. Ecology of Freshwater Fish 2: 116–120.
Last, P.R., Scott, E.O.G. & Talbot, F.H. 1983. Fishes of Tasmania. Hobart : Tasmanian Fisheries Development Authority 563 pp. figs.
Macey, D.J., Cake, M.H. & Potter, I.C. 1988. Exceptional iron concentrations in larval lampreys (Geotria australis) and the activities of superoxide radical detoxifying enzymes. Biochemical Journal 252(1): 167-172.McDowall, R.M. 1988. Diadromy in fishes: migrations between freshwater and marine environments. Croom Helm, London.
McDowall, R.M. (ed.) 1996. Freshwater Fishes of South-eastern Australia. Sydney : Reed Books 247 pp.
McDowall, R.M. & Stewart, A.L. 2015. 3 Family Geotriidae. pp. 35-36 in Roberts, C.D., Stewart, A.L. & Struthers, C.D. (eds). The Fishes of New Zealand. Wellington : Te Papa Press Vol. 2 pp. 1-576.
Merrick, J. & Schmida, G. 1984. Australian Freshwater Fishes. Macquarie University, North Ryde, NSW.
Morgan, D.L., Gill, H.S. & Potter, I.C. 1998. Distribution, identification and biology of freshwater fishes in south-western Australia. Records of the Western Australian Museum Supplement 56: 1-97.
Neira, F.J., Bradley, J.S., Potter, I.C. & Hillard, R.W. 1988. Morphological variation among widely dispersed larval populations of anadromous southern hemisphere lampreys (Geotriidae and Mordaciidae). Zoological Journal of the Linnean Society 92: 383-408
Ogilby, J.D. 1896. A monograph of the Australian Marsipobranchii. Proceedings of the Linnean Society of New South Wales 21(3): 388-426 (as Velasia stenostomus)
Potter, I.C. 1986. The distinctive characters of southern hemisphere lampreys (Geotriidae and Mordaciidae). pp. 9-29 in Uyeno, T., Arai, R., Taniuchi, T. & Matsuura, K. (eds). Indo-Pacific Fish Biology. Proceedings of the Second International Conference on Indo-Pacific Fishes. Tokyo : Ichthyological Society of Japan 985 pp.
Potter, I.C., Hilliard, R.W. & Bird, D.J. 2009. Metamorphosis in the Southern Hemisphere lamprey, Geotria australis. Journal of Zoology 190: 405–430.
Potter, I.C., Hilliard, R.W. & Bird, D.J. & Macey, D.J. 1983. Quantitative data on morphology and organ weights during the protracted spawning-run period of the Southern Hemisphere lamprey Geotria australis. Journal of Zoology 200: 1-20.
Potter, I.C., Macey, D.J., Roberts, A.R. & Withers, P.C. 1996. Oxygen consumption by ammocoetes of the lamprey Geotria australis in air. Journal of Comparative Physiology B: Biochemical, Systemic, and Environmental Physiology 166(5): 331-336.
Potter, I.C., Prince, P & Croxall, J. 1979. Data on the adult marine and migratory phases in the life cycle of the southern hemisphere lamprey Geotria australis. Environ Biol Fishes 4: 65–69.
Potter, I.C. & Strahan, R. 1968. The taxonomy of lampreys Geotria and Mordacia and their distribution in Australia. Proceedings of the Linnean Society of London 179(2): 229-240 figs 1-2.
Raadik, T.A. 1992. Distribution of freshwater fishes in east Gippsland, Victoria, 1967–1991. Proceedings of the Royal Society of Victoria 104: 1-22
Renaud, C.B. 2011. Lampreys of the World. An Annotated and Illustrated Catalogue of Lamprey Species Known to Date. FAO Species Catalogue for Fisheries Purposes No. 5. Rome : FAO 116 pp.
Renaud, C.B., Gill, H.S. & Potter, I.C. 2009. Relationships between the diets and characteristics of the dentition, buccal glands and velar tentacles of the adults of the parasitic species of lamprey. Journal of Zoology 278: 231-242.
Robinson, E.S. & Potter, I.C. 1981. The chromosomes of the southern hemispheric lamprey, Geotria australis Gray. Experientia 37(3): 239-240.
Salas, C.A., Yopak, K.E., Warrington, R.E., Hart, N.S., Potter, I.C. & Collin, S.P. 2015. Ontogenetic shifts in brain scaling reflect behavioral changes in the life cycle of the pouched lamprey Geotria australis. Frontiers in Neuroscience 9: 251. https://doi.org/10.3389/fnins.2015.00251
Schwab, I.R. & Collin, S.P. 2005. Are you calling me primitive? British Journal of Ophthalmology 89: 1553.
Scott, T.D., Glover, C.J.M. & Southcott, R.V. 1974. The Marine and Freshwater Fishes of South Australia. Adelaide : Government Printer 392 pp. figs.
Strahan, R. 1959. The status of Yarra singularis and Geotria australis (Petromyzonidae). Journal of the Royal Society of Western Australia 42: 49-52
Strahan, R. 1960. A comparison of the ammocoete and macrophthalmia stages of Mordacia mordax and Geotria australis (Petromyzonidae). Pacific Science 14: 416-420
Strahan, R. 1980. Family Mordaciidae: short-headed lampreys; Family Geotriidae: pouched lamprey. pp. 38-43 6 figs in McDowall, R.M. (ed.) Freshwater Fishes of South-eastern Australia. Sydney : A.H. & A.W. Reed 208 pp. figs
Waite, E.R. 1921. Illustrated catalogue of the fishes of South Australia. Records of the South Australian Museum (Adelaide) 2(1): 1-208 293 figs pl. 1
Waite, E.R. 1923. The Fishes of South Australia. Adelaide : Government Printer 243 pp. 325 figs.
Whitley, G.P. 1932. The lancelets and lampreys of Australia. The Australian Zoologist 8(3): 256-264 4 figs pl. 13
Whitley, G.P. 1940. The Fishes of Australia. Part 1. The sharks, rays, devil-fish, and other primitive fishes of Australia and New Zealand. Sydney : Roy. Zool. Soc. N.S.W. 280 pp. 303 figs.
Whitley, G.P. 1956. Freshwater fishes of Australia. 2. — Lampreys. Aqualife 1(9): 9-12
Yamazaki, Y. & Goto, A. 2000. Present status and perspectives on the phylogenetic systematics and speciation of lampreys. Japanese Journal of Ichthyology 47(1): 1-28.