- Classification
- ACTINOPTERYGII
- SYNGNATHIFORMES
- SYNGNATHIDAE
-
Fish Classification
-
Class
ACTINOPTERYGII Ray-finned fishes -
Order
SYNGNATHIFORMES Seahorses, pipefishes -
Family
SYNGNATHIDAE Seahorses, pipefishes -
Genera
Acentronura(1) Bhanotia(1) Bulbonaricus(2) Campichthys(3) Choeroichthys(5) Corythoichthys(8) Cosmocampus(4) Doryrhamphus(3) Dunckerocampus(3) Festucalex(3) Filicampus(1) Halicampus(8) Haliichthys(1) Heraldia(1) Hippichthys(5) Hippocampus(28) Histiogamphelus(2) Hypselognathus(2) Idiotropiscis(3) Kaupus(1) Kimblaeus(1) Leptoichthys(1) Lissocampus(3) Maroubra(1) Micrognathus(4) Microphis(2) Mitotichthys(4) Nannocampus(2) Notiocampus(1) Penetopteryx(1) Phoxocampus(2) Phycodurus(1) Phyllopteryx(2) Pugnaso(1) Siokunichthys(1) Solegnathus(6) Stigmatopora(4) Stipecampus(1) Syngnathoides(1) Trachyrhamphus(2) Urocampus(4) Vanacampus(4)
Family SYNGNATHIDAE
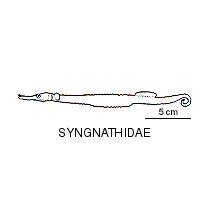
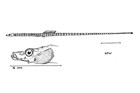
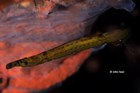
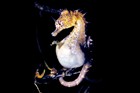
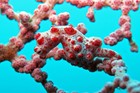
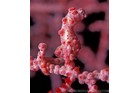
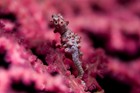
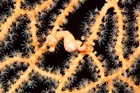
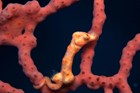
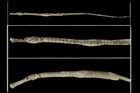
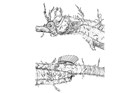
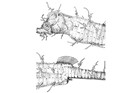
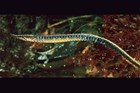
A large and diverse group of pipefishes, seahorses, seadragons and pipehorses, all having a tiny mouth at the end of a tubular snout and semi-flexible bodies encased in bony rings.
Syngnathids lack fin spines and pelvic fins, and other fins may be reduced or absent. They have a unique reproductive mode where the female deposits her eggs into an abdominal brood pouch or onto a modified exposed region on the underside of her male partner. The male then incubates and nourishes the developing embryos until they hatch.
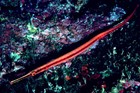
These extraordinary cryptic fishes are often ornamented with filaments and other appendages to enhance their excellent camouflage.
More Info
|
Family Taxonomy |
A large family with more than 340 recognised species in more 50 genera. In Australian waters about species in 40 genera are recognised. |
|
Family Distribution |
Syngnathids are found worldwide in marine tropical to temperate waters, mostly in depths above 50 m. Some species are trawled in deeper shelf and upper slope waters, and several live in estuarine and freshwater environments. Syngnathids are mostly benthic on coastal reefs, amongst marine algae and seagrass beds, or on sandy and rubble substrates and in caves and crevices. A few species are found offshore amongst floating Sargassum algae. |
|
Family Description |
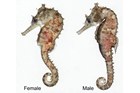
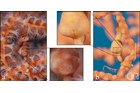
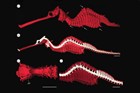
Meristic features: Dorsal fin 7-64; Anal fin 0-6; Pectoral fin 0-26; Caudal 0-11; Body rings: abdominal 8-28 + caudal 14-91; Branchiostegal rays 1-3; Circumorbital bones 2-3. Body typically elongate, mouth very small at end of a tubular snout; gill opening a small pore in the opercular membrane. Sygnathids lack fin spines and pelvic fins; other fins are variously present, absent or reduced. They are encased in a series of bony rings. Seahorses differ from most other syngnathids in having the head at a 90° angle to the body axis and a distinctly prehensile tail with which they cling to the substrate. |
|
Family Size |
Range in size from less than 10 mm (pygmy seahorses) to about 65 cm, although most species are relatively small. |
|
Family Colour |
Variable in colour pattern and body ornamentation. Most species are cryptically coloured to match their surroundings and many species are adorned with dermal appendages and filaments. Most are extremely well camouflaged, often mimicing seagrasses and algae in their environment. |
|
Family Feeding |
Carnivores, feeding primarily on small benthic and pelagic crustaceans, including copepods, amphipods, mysids and shrimps. Most syngnathids feed by rapidly and powerfully sucking prey items in through their long snouts. Syngnathids are preyed upon by a wide variety of predators ranging from cephalopods to sharks, rays, bony fishes, sea birds, turtles and marine mammals. |
|
Family Reproduction |
The sexes are separate and fertilisation is external. Males and females often live in pairs and may occupy small home ranges. Many seahorse and pipefishes form monogamous pair bonds and some perform daily greeting rituals during the breeding season. Females deposit their eggs in a brood pouch, or onto a modified brood area on the underside of the male. The eggs are then fertilised by the male who then incubates and nourishes the developing embryos until the young hatch. Syngnathid eggs are generally spherical except in Hippocampus, which has ellipsoidal or pear-shaped eggs. The larvae are planktonic, and postlarvae resemble miniature adults. Juveniles often remain in the plankton for extended periods. |
|
Family Commercial |
Most species are of no commercial importance, however, over-fishing has led to the decline of some shallow water species. Seahorses in particular are traded worldwide for use in the Traditional Chinese Medicine (TCM) industry and as popular aquarium fishes, or are simply dried and sold as curios. A number of species are bred in captivity in for trade in the aquarium industry. Although Australia mainly exports live syngnathids for the aquarium trade, a few kilos of dried sygnathids are exported to Asia. In Australia, strong legislation at both Commonwealth (national) and State levels protect syngnathids and/or require monitoring of their exploitation. All exports of syngnathids from Australia require permits under approved management plans (Vincent et al. 2011b) |
|
Family Conservation |
All Hippocampus species and some other syngnathids are on the IUCN Red List of Threatened Species. In 2004, Seahorses were listed on Appendix II of CITES, the Convention on International Trade in Endangered Species of Wild Fauna and Flora. In those countries which are signatory to the Convention, the export of seahorses must not be detrimental to wild seahorse populations. All sygnathid species are listed in Australia as threatened under the Environment Protection and Biodiversity Conservation Act 1999 EPBC Act. Various State Government agencies protect syngnathid species. Syngnathids are protected under the Victorian Fisheries Act 1995, the Tasmania Living Marine Resources Management Act 1995 and the South Australian Fisheries Management Act 2007. |
|
Family Remarks |
The fossil record of syngnathids dates back to the Eocene, and the oldest known fossil pipefishes are from Monte Bolca in Italy. In 2005, Miocene seahorse fossils were found in the Tunjice Hills of Slovenia. |
|
Family Biology |
|
|
Author |
Bray, D.J. 2021 |
|
Family Resources |
References
Allen, G.R. 1997. Marine Fishes of Tropical Australia and South-east Asia. Western Australian Museum, Perth.
Ashley-Ross, M.A. 2002. Mechanical properties of the dorsal fin muscle of seahorse (Hippocampus) and pipefish (Syngnathus). J. Exp. Zool. 293: 561-577.
Berglund, A. & Rosenqvist, G. 1993. Selective males and ardent females in pipefishes. Behav. Ecol. Sociobiol. 32: 331–336.
Berglund, A. & Rosenqvist, G. 2003. Sex role reversal in pipefish. Adv. Stud. Behav. 32: 131–167.
Breder, C.M. & D.E. Rosen. 1966. Modes of reproduction in fishes. T.F.H. Publications, Neptune City, New Jersey. 941 p.
Bruner, E. & V. Bartolino. 2008. Morphological variation in the seahorse vertebral system. Int. J. Morphol. 26(2): 247-262.
Casey, S.P., H.J. Hall, H.F. Stanley & A.C.J. Vincent. 2004. The origin and evolution of seahorses (genus Hippocampus): a phylogenetic study using the cytochrome b gene of mitochondrial DNA. Mol. Phylo. Evol. 30: 261-272.
Carcupino, M., A. Baldacci, M. Mazzini & P. Franzoi. 2002. Functional significance of the male brood pouch in the reproductive strategies of pipefishes and seahorses: a morphological and ultrastructural comparative study on three anatomically different pouches. J. Fish Biol. 61: 1465-1480.
Clutton-Brock, T.H. & Vincent, A.C.J. 1991. Sexual selection and the potential reproductive rates of males and females. Nature 351: 58–60.
Consi, T.R., P.A. Seifert, M.S. Triantafyllou & E.R. Edelman. 2001. The dorsal fin engine of the seahorse (Hippocampus sp.) J. Morphol. 248: 80-97.
Dawson, C.E., 1984 A new pipehorse (Syngnathidae) from western Australia, with remarks on the subgenera of Acentronura. Japan. J. Ichthyol. 31(2): 156-160.
Dawson, C.E. 1985. Indo-Pacific Pipefishes (Red Sea to the Americas). Gulf Coast Research Laboratory, Ocean Springs (Mississippi), 230 pp., 239 figs.
Foster, R. & M.F. Gomon. 2010. A new seahorse (Teleostei: Syngnathidae: Hippocampus) from south-western Australia. Zootaxa 2613: 61-68.
Foster, S.J. & A.C.J. Vincent. 2004. Life history and ecology of seahorses: implications for conservation and management. J. Fish Biol. 65: 1-61.
Foster, S.J. & A.C.J. Vincent. 2005. Enhancing sustainability of the international trade in seahorses with a single minimum size limit. Conservation Biology 19: 1044-1050.
Fritzsche, R.A. 1984. Gasterosteiformes: Development and Relationships. In Moser, H.G., W.J. Richards, D.M. Cohen, M.P. Fahay, A.W. Kendall, Jr. & S.L. Richardson (eds.) Ontogeny and Systematics of Fishes. Special Publication no. 1, American Society of Ichthyologists and Herpetologists, Lawrence, KS.
Giles, B.G., Ky, T.S., Hoang, D.H. & Vincent, A.C.J. 2006. The catch and trade of seahorses in Vietnam. Biodiversity and Conservation 15: 2497-2513.
Gomon, M.F. & F.J. Neira. 1998. Syngnathidae: pipefishes and seahorses. p. 122-131. In F.J. Neira, F.J., A.G. Miskiewicz & T. Trnski (eds.) Larvae of temperate Australian fishes: laboratory guide for larval fish identification. University of Western Australia Press. 474 pp.
Hale, M. E. 1993. Mechanisms of bending and holding in seahorses and pipefishes (Teleostei: Syngnathidae). Am. Zool. 33: 120A.
Hamilton, H., Saarman, N., Short, G., Sellas, A.B., Moore, B., Hoang, T., Grace, T.C.L., Gomon, M., Crow, K. & Simison, W.B. 2016. Molecular phylogeny and patterns of diversification in syngnathid fishes. Molecular Phylogenetics and Evolution 107: 388-403 + supplement 1-4 + 5. https://doi.org/10.1016/j.ympev.2016.10.003
Harasti, D., Glasby, T.M. & Martin-Smith, K.M. 2010. Striking a balance between retaining populations of protected seahorses and maintaining swimming nets. Aquatic Conservation: Marine and Freshwater Ecosystems 20: 159-66.http://HippocampusInfo.org/
Hoffman, E.A., K.B. Mobley & A.G. Jones. 2006. Male pregnancy and the evolution of body segmentation in seahorses and pipefishes. Evolution 60: 404-410.
IUCN List of Threatened Species http://www.iucnredlist.org/search/
Jones, A.G. & Avise, J.C. 2001. Mating systems and sexual selection in male-pregnant pipefishes and seahorses: insights from microsatellite-based studies of maternity. J. Hered. 92: 150-158.
Jones, A.G., Moore, G.I., Kvarnemo, C., Walker, D. & Avise, J. 2003. Sympatric speciation as a consequence of male pregnancy in seahorses. PNAS 100(11): 6598-6603.
Kawahara, R., M. Miya, K. Mabuchi, S. Lavoue, J.G. Inoue, T.P. Satoh, A. Kawaguchi & M. Nishida. 2008. Interrelationships of the 11 gasterosteiform families (sticklebacks, pipefishes, and their relatives): A new perspective based on mitogenome sequences from 75 higher teleosts. Mol. Phylogenet. Evol. 46(1): 224-236.
Kendrick, A.J. & G.A. Hyndes. 2003. Patterns in the abundance and size-distribution of syngnathid fishes among habitats in a seagrass-dominated marine environment. Estuarine Coastal and Shelf Science 57(4): 631-640.
Kendrick, A.J. & G.A. Hyndes. 2005. Variations in the dietary compositions of morphologically diverse syngnathid fishes. Env. Biol. Fishes 72(4): 415-427.
Kirby, R. R., Johns, D. G. & Lindley, J. A. 2006. Fathers in hot water: rising sea temperatures and northeastern Atlantic pipefish baby boom. Biol. Lett. 2: 597-600.
Kleiber, D., L.K. Blight & I.R. Caldwell & A.C.J. Vincent. 2010. The importance of seahorses and pipefishes in the diet of marine animals. Rev Fish Biol Fisheries 21(2): 205-223.
Koldewey, H.J. & K.M. Martin-Smith. 2010. A global review of seahorse aquaculture. Aquaculture 32: 131-152.
Kuiter, R.H. 1996. Guide to Sea Fishes of Australia. New Holland Press, Sydney. 433 pp. figs.
Kuiter, R.H. 2001. Revision of the Australian Seahorses of the genus Hippocampus (Syngnathiformes: Syngnathidae) with descriptions of nine new species. Rec. Aust. Mus. 53: 239-340.
Kuiter, R.H. 2004 A new pygmy pipehorse (Pisces: Syngnathidae: Idiotropiscis) from Eastern Australia. Rec. Aust. Mus. 56: 162-165.
Kuiter, R.H. 2008. Family Syngnathidae (pp. 448-479). In Gomon, M.F., D.J. Bray & R.H. Kuiter. (Eds.) Fishes of Australia's Southern Coast. New Holland Press & Museum victoria, Melbourne, Australia, 928 pp.
Kuiter, R.H. 2009. Seahorses and their relatives. Aquatic Photographics, Seaford, Australia. 333 p.
Kvarnemo, C. & L.W. Simmons. 2004. Testes investment and spawning mode in pipefishes and seahorses (Syngnathidae). Biol. J. Linn. Soc. 83: 369-376.
Longo, S.J., Faircloth, B.C., Meyer, A., Westneat, M.W., Alfaro, M.E. & Wainwright, P.C. 2017. Phylogenomic analysis of a rapid radiation of misfit fishes (Syngnathiformes) using ultraconserved elements. Molecular Phylogenetics and Evolution 113: 33-48, https://doi.org/10.1016/j.ympev.2017.05.002.
Lourie, S.A., A.C.J. Vincent & H.J. Hall 1999. Seahorses. An identification guide to the world's species and their conservation. Project Seahorse, 214 pp.
Martin-Smith, K.M., Vincent, A.C.J. 2006. Exploitation and trade of Australian seahorses, pipehorses, sea dragons and pipefish (Family Syngnathidae). Oryx 40: 141-151.
Mobley, K.B., C.M. Small & A.G. Jones. 2011. The genetics and genomics of Syngnathidae: pipefishes, seahorses and seadragons. Journal of Fish Biology 78: 624–1646.
Monteiro, N.M., Almada, V.C. & Vieira, M.N. 2005. Implications of different brood pouch structures in syngnathid reproduction. J. Mar. Biol. Assoc. UK 85: 1235-1241.
Nelson, J.S. 2006. Fishes of the World. 4th Ed. John Wiley & Sons, Inc., Hoboken, New Jersey. 601 pp.
O’Donnell, K.P., M.G. Pajaro & A.C.J. Vincent. 2010. How does the accuracy of fisher knowledge affect seahorse conservation status? Animal Conservation 13(6): 526-523.
Orr, J.W. & T.W. Pietsch. 1994. Pipefishes and their allies. In Paxton, J.R. & W.N. Eschmeyer (eds.) Encyclopedia of Fishes. Academic Press, San Diego.
Paulus, T. 1999. Family Syngnathidae, pp. 2264-2276 In Carpenter, K.E. & V.H. Niem (eds.) The Living Marine Resources of the Western Central Pacific. FAO Species Identification Guide for Fisheries Purposes. FAO, Rome, Vol 4.
Paxton, J. R., J. E. Gates, D. F. Hoese & D. J. Bray. 2006. Syngnathidae (Pp. 810-846). In Beesley, P.L. & Wells, A. (Eds) Zoological Catalogue of Australia. Volume 35. Fishes. ABRS & CSIRO Publishing, Australia., 3 vols.
Pogonoski, J.J., D.A. Pollard & J.R. Paxton. 2002. Conservation Overview and Action Plan for Australian Threatened and Potentially Threatened Marine and Estuarine Fishes. Environment Australia, Canberra. 375 pp.
Project Seahorse http://seahorse.fisheries.ubc.ca/index.html
Ripley, J.L. & C.M. Foran. 2006. Placental transfer - its not just for mammals anymore. Mar. Environ. Res. 62: S268-S269.
Randall, J.E., G.R. Allen & R.C. Steene. 1997. Fishes of the Great Barrier Reef and Coral Sea. University of Hawaii Press, Honolulu.
Rosenqvist, G. & A. Berglund. 2011. Sexual signals and mating patterns in Syngnathidae. Journal of Fish Biology 78: 1647–1661.
Sanders, J.G., Cribbs, J.E., Feinberg, H.G., Hulburd, G.C., Katz, L.S. & Palumbi, S.R. 2008: The tip of the tail: molecular identification of seahorses for sale in apothecary shops and curio stores in California. Conservation Genetics 9: 65-71.
Scales, H. 2010. Advances in the ecology, biogeography and conservation of seahorses (genus Hippocampus). Progress in Physical Geography April 2010: 1-19.
Schmid, M.S. & D.G. Senn. 2002. Seahorses - Masters of adaptation. Vie Et Milieu-Life and Environment 52: 201-207.
Shokri, M.R., W. Gladstone & J. Jelbart. 2009. The effectiveness of seahorses and pipefish (Pisces: Syngnathidae) as a flagship group to evaluate the conservation value of estuarine seagrass beds. Aquatic Conservation: Marine and Freshwater Ecosystems 19: 588-595.
Stolting, K.N. & Wilson, A.B. 2007. Male pregnancy in seahorses and pipefish: beyond the mammalian model. BioEssays 29: 884-96.
Teske, P.R., M.I. Cherry & C.A. Matthee. 2004. The evolutionary history of seahorses (Syngnathidae: Hippocampus): molecular data suggest a West Pacific origin and two invasions of the Atlantic Ocean. Molecular Phylogenetics and Evolution 30: 273-286.
Teske, P.R., H. Hamilton, P.J. Palsboll, C.K. Choo, H. Gabr, S.A. Lourie, M. Santos, A. Sreepada, M.I. Cherry & C.A. Matthee. 2005. Molecular evidence for long-distance colonization in an Indo-Pacific seahorse lineage. Marine Ecology-Progress Series 286: 249-260.
Teske, P.R., Hamilton, H., Matthee, C.A. & Barker, N.P. 2007. Signatures of seaway closures and founder dispersal in the phylogeny of a circumglobally distributed seahorse lineage. BMC Evol. Biol. 7: 138.
Teske, P. & L. Beheregaray. 2009. Evolution of seahorses' upright posture was linked to Oligocene expansion of seagrass habitats. Biology Letters 5(4): 521-523
Teske, P. & L. Beheregaray. 2009. The rise of seahorses. Australian Science Nov/Dec: 24-25.
Vari, R.P. 1982. Seahorses (Subfamily: Hippcampinae). Fishes of the Western North Atlantic. Memoirs of the Sears Foundation for Marine Research 1(8): 173-189.
Vincent, A.C.J. 1994. Seahorses exhibit conventional sex roles in mating competition, despite male pregnancy. Behaviour 128: 135-151.
Vincent, A.C.J. 1996. The International Trade on Seahorses. Cambridge, UK : Traffic, International.
Vincent, A., I. Ahnesjö, A. Berglund & G. Rosenqvist. 1992. Pipefishes and seahorses: are they all sex role reversed? Trends Ecol. Evol. 7: 237-241.
Vincent, A.C.J., Berglund, A. & I. Ahnesjo. 1995. Reproductive ecology of five pipefish species in one eelgrass meadow. Environmental Biology of Fishes 44: 347-361.
Vincent, A.C.J., Foster, S.J. & Koldewey, H.J. 2011. Conservation and management of seahorses and other Syngnathidae. Journal of Fish Biology. 78(6): 1681-1724
Vincent, A.C.J. & L.M. Sadler. 1995. Faithful Pair Bonds in Wild Seahorses, Hippocampus whitei. Animal Behaviour 50: 1557-1569.
Wilson, N.G. & Rouse, G.W. 2010. Convergent camouflage and the non-monophyly of ‘seadragons’ (Syngnathidae: Teleostei): suggestions for a revised taxonomy of syngnathids. Zoologica Scripta 39: 551-558.
Wilson, T.B., I. Ahnesjö, A.C.J. Vincent & A. Meyer. 2003. The dynamics of male brooding, mating patterns, and sex roles in pipefishes and seahorses (Family Syngnathidae). Evolution 57: 1374-1386.
Wilson, A.B. & J.W. Orr. 2011. The evolutionary origins of Syngnathidae: pipefishes and seahorses. Journal of Fish Biology 78: 1603–1623.
Wilson, T.B., A. Vincent, I. Ahnesjö & A. Meyer. 2001. Male pregnancy in seahorses and pipefishes (Family Syngnathidae): Rapid diversification of paternal brood pouch morphology inferred from a molecular phylogeny. Journal of Heredity 92: 159-166.
Zalohar, J., Hitij, T. & Kriznar, M. 2009. Two new species of seahorses (Syngnathidae, Hippocampus) from the Middle Miocene (Sarmatian) Coprolitic Horizon in Tunjice Hills, Slovenia: the oldest fossil record of seahorses. Ann. Paléontol. 95(2): 71-96.
Zhang, Y-H., Ravi, V., Qin, G., et al. 2020. Comparative genomics reveal shared genomic changes in syngnathid fishes and signatures of genetic convergence with placental mammals, National Science Review 7(6): 964–977, https://doi.org/10.1093/nsr/nwaa002